Human Microbiota

From the series ‘Micropolis’, Max Meyer, 2014.
It was unseasonably cold and I was bundled up to keep myself warm as I walked from the transit stop to the lab. My friend told me that they’d meet me there at 6:30 and I was running a little bit late. They met me by the door and guided me past several keycard-locked doors to their laboratory. It was a massive, cluttered room, and it was nearly empty. Every surface bristled in Pyrex glassware; large, translucent, variously labeled plastic jugs; binders, notes, diagram; or boxes upon boxes of gloves. My friend sat me down while we waited for the autoclave to finish sterilizing the equipment we’d be using.
They left me to my own devices for a few minutes and went to putter about, attending to their own ongoing work, but not before cautioning me not to touch anything, especially not any door handles. I wandered about and looked at the posters from various conferences with which people had decorated the space. I paused and scrutinized post-its stuck to cubby walls, trying to recall my last experiences with chemistry or biology — nearly a decade ago — in order to wring some meaning from the notations and formulae.
Once everything was ready, I struggled into a pair of size large nitrile gloves (apparently the people who use this lab skew smaller) and was ushered into a small room. I was directed to something that looked like a fume hood, but functioned in reverse, blowing a steady stream of sterilized air over the work surface. Following my friend’s instruction, I carefully poured the blood-warm maroon agar preparation into ten petri dishes, and while we waited to solidify, they talked me through the basics. These (small, individually wrapped, flexible plastic sticks) are inoculation loops, you’ll be using those to collect the samples. This (gently dragging the sample-laden tip of the inoculation loop back and forth in each quarter of the petri dish) is the traditional streaking method. This (looks like a Sharpie) is a marker.
I quickly ran through the list of places on my body I was hoping to sample. It felt very personal to be sharing this room with a friend while trying to collect microbes that lived on and in my body, but they didn’t want to leave me unattended in the lab, so I just swallowed the awkwardness and began collecting. After having twice surreptitiously slid an inoculation loop down the front of my pants, I lost my nerve dragged the inoculation loop across the bottom of my right foot, rather than remove my sock entirely and in an effort to tap that rich vein between my toes.
The dishes hopefully laden with countless thousands of lifeforms, my friend expertly sealed them up with a plasticky tape. They told me that e. coli doubles every eighteen minutes, so, if I could keep the colonies warm, I should be able to see some results in a few days. As I left the lab, I tucked the small box containing parts of myself into my coat to keep my microscopic charges safe from the sub-zero temperatures.

From the series ‘Micropolis’, Max Meyer, 2014.
Each of us is made up of roughly one hundred trillion cells, give or take a few hundred million. Of those, only ten percent are human; the remaining ninety percent consists of hundreds of species of bacteria, viruses, mites and fungi (Grice & Segre, 2011, p. 244.). Despite having a dominant numerical majority, these teeming hordes are vastly smaller than the cells that we think of as human, as components of ourselves. All told, these 1014 microbes weigh roughly one to two kilograms (O’Keefe, 2008, p. 51.). Compared to the rest of our body, this may seem simultaneously significant and insignificant. Sure, a couple kilograms is pretty minor compared to the rest of our body, but on the other hand there’s an invisible community of foreign species seething around and through your body that weighs about as much as your brain.
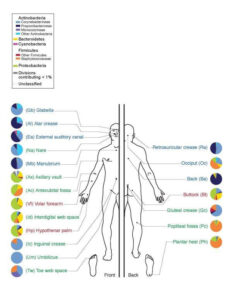
These invisible entities bear only their Linnaean names, and these do not conform readily to the eye or tongue: Staphylococcus, Propionibacteria, Micrococcus on the skin (“Skin Flora,” n.d.) or Prevotella, Bacteroides and Ruminococcus in the gut (“Gut Flora,” n.d.). From their lack of common names alone it seems clear that these are not forms of life of which most people regularly think; nobody rides an Equus ferus caballus and, with the help of their trusty Canis lupus familiaris corrals a herd of Capra aegagrus hircus. The microbes whose names have become part of common parlance are invariably those of which we are afraid. We don’t speak of bacteriophages in the mouth acting as predators to regulate and sustain a complex oral microecosystem, we hear about people becoming seriously ill from MRSA (Methicillin-resistant Staphylococcus aureus); we don’t consider the bacteria that synthesize vitamin K2 (Bentley & Meganathan, 1982.), we fret over the presence of Escherichia coli or Salmonella in our food. This is not to discount the dangers presented by pathogenic microbes like those I have just mentioned, but rather to argue for a more holistic view of microbial life than thinking of it simply as representative of the unclean and a harbinger of disease.
My schedule was such that I did not have time to bring my microscopic charges home and get them settled in a nice warm place. So I kept fidgeting with them under my coat, making sure there weren’t any cracks where possibly dangerous cold winds could sneak in. Though I felt protective of these little colonies, I simultaneously felt as though I was doing something wrong, something transgressive. These petri dishes contained bacteria. I was growing them. On purpose. The fact that I, and everyone else that I passed by on the street, was swarming with vital populations which dwarfed my fledgling colonies was immaterial. These were gross objects and walking around with them in public was an antisocial act. I had better keep these things hidden or risk the consequences.
It’s incredibly easy to ignore something that we cannot sense directly, even if this particular something happens to be ubiquitously integrated with our corporeal selves. I contend that there is more at work here than mere sensory limitation; as a species we have a wealth of knowledge about countless forms of life and phenomena that are either too small, distant, or complex for us to engage with directly, yet knowledge of our corporeal microbiomes is a field that has only recently begun to be researched in earnest. In 2008 the US’s National Institute of Health began the Human Microbiome Project (HMP) in an attempt to better understand the role of our microbiome in sustaining health (“Human Microbiome Project”, n.d.).The HMP was a significant break from previous methods of investigating the inhabitants in our bodily ecosystems because the development of new devices allowed researchers to sample DNA directly from microbes in their natural habitat (Bourzac, 2007.). The older method was to take samples from sites on the body and then culture them in petri dishes. However, such techniques “essentially select for laboratory ‘weeds’: species that flourish under the typical nutritional and physiological conditions that are used by diagnostic microbiology laboratories. These are not necessarily the most abundant or influential organisms in the community” (Grice & Segre, 2011, p. 247).
The method of research used by the HMP is known as metagenomics, a process by which “…DNA sequence information is extracted from entire microbial communities in situ. Metagenomic approaches use this bulk data to infer underlying properties of both individual microbes and microbial communities as a whole” (DeLong, 2007, p. 26.). Metagenomics has yielded a staggering amount of new information about our personal ecosystems and has shown that there is a huge amount of heterogeneity between different sites on the body and also a remarkable amount of variation between individual humans (Schloss, 2014, p. 44.).
With this new, ever-broadening picture of what the human microbiome looks and works like, come challenging questions. How do we better interact with our microbial populations for our mutual benefit? Will our new understanding of our bodies as ecosystems impact how we engage with our environmental macroecosystems? How can we still be thought of as individuals?

From the series ‘Micropolis’, Max Meyer, 2014.
So, if it’s not just a physical limitation brought on by the simple technical problems of knowing something so small, then why might such broad swaths of our microbiome still be terra incognita? In his Complete Psychological Works, Freud argues that
civilized men are clearly embarrassed by anything that reminds them too much of their animal origin. […] Since however, the must necessarily remain far removed from such perfection, men have chosen to evade the predicament by so far as possible denying the very existence of the this inconvenient “trace of the Earth” by concealing it from one another, and by withholding it from the attention and care which it might claim as an integrating component of their essential being. The wiser course would undoubtedly have been to admit its existence and to dignify it as much as its nature will allow. (qtd in Rosebury, 1969, p. 57)
This florid passage dovetails nicely with the frequency of ritual cleansing and ablution throughout most major religions. Judaism, Sikhism and Islam all have specific indications for hand and body hygiene (World Health Organization, 2009.), hinting at the one of the possible origins of the emotional link many people feel between hygiene and propriety. Again, I do not seek to challenge the importance of hygiene, or the validity of any of these religious practices, but fear that our pursuit of cleanliness and subsequent ignorance of the nature of our bodies as complex, individual ecosystems may be finally manifesting substantial consequences.

From the series ‘Micropolis’, Max Meyer, 2014.
There are many similarities between how we think of our microbiome and how we interact with our macrobiome: we don’t consider it at all until something goes wrong. Drawing upon the work of Giorgio Agamben, Smith points to human exemptionalism and exceptionalism — two epistemologies that inform our interactions with other life on Earth — as the causes of our willful detachment from our surroundings (Smith, 2013, p. 23-24.). Humanity’s bad habit of thinking itself as separate from and superior to other forms of life has resulted in resource depletion, climate change, and mass extinctions. Much as these problems have brought about environmental movements, pressures to develop sustainable energy solutions and increased interest in conservation, abuse of our microbes may be manifesting itself as a variety of health problems and said problems are some of the raisons d’être for these new forays into our microbiome.
In 1989, David Strachan undertook a study which compared the prevalence of hay fever to several social and environmental factors. The factors which had the strongest inverse correlation were family size and order of birth; the larger the family and the more older siblings one is born with, the less likely one is to develop hay fever (Strachan, 1989, p. 1259.). This study was integral to the development of the hygiene hypothesis: that exposure to a variety of microbial life (from a number of older siblings, say) in childhood trains the immune system to react appropriately to various foreign material; pathogenic microbes will be met with a full defensive reaction, but other substances, potential allergens or the body’s own cells — in the case of autoimmune disorders like asthma, Crohn’s disease and multiple sclerosis — would be left unmolested (Okada, Kuhn, Feillet, Bach, 2010, p. 2.).
From 1998 to 2008, there has been a significant increase in many forms of immunological disorders, from asthma and atopic dermatits to type 1 diabetes and ulcerative colitis (Okada et al., 2010, p. 1.). Studies have shown that there is a correlation between excessive cleanliness and immunological disruption: either in the form of susceptibility to pathogenic  microbes or developing autoimmune disorders, although improved hygiene and sanitation has reduced the incidence of infectious disease (Bloomfield, Stanwell-Smith, Crevel, Pickup, 2006, p. 407.). This begs the question, how clean is clean enough? As yet, there is no clear answer to this question, but some hygienic practices seem to be based more on emotion and marketing than biological necessity. For example, hand sanitizer use can be somewhat fraught. It only works better than handwashing with soap in certain circumstances, and non-alcohol-based hand sanitizers may result in the microbial populations being reduced and developing resistance to the sanitizers (Center for Disease Control and Prevention, 2014.). Worse yet, the use of some popular antimicrobial agents may have wide-ranging health implications. One such compound, triclosan, may be of limited use in household products it has been used in — soaps, deodorants, shaving creams, cosmetics and dishwashing liquids — and it is not removed during waste water treatment (Schwarcz, 2014.). Triclosan has been in wide use for forty years, and traces can be found in the urine of a majority of the US population (Calafat, Ye, Wong, Reidy, Needham, 2007.). Ironically, this product, intended to protect our health, may be jeopardizing it by disrupting the human microbiome, to say nothing of the effects of triclosan on fish and other aquatic life (Schwarcz, 2014.). Many governments are contemplating banning the substance (CBC News, 2014.), but this is but one example of our fear and ignorance of microbial life gone awry.
microbes or developing autoimmune disorders, although improved hygiene and sanitation has reduced the incidence of infectious disease (Bloomfield, Stanwell-Smith, Crevel, Pickup, 2006, p. 407.). This begs the question, how clean is clean enough? As yet, there is no clear answer to this question, but some hygienic practices seem to be based more on emotion and marketing than biological necessity. For example, hand sanitizer use can be somewhat fraught. It only works better than handwashing with soap in certain circumstances, and non-alcohol-based hand sanitizers may result in the microbial populations being reduced and developing resistance to the sanitizers (Center for Disease Control and Prevention, 2014.). Worse yet, the use of some popular antimicrobial agents may have wide-ranging health implications. One such compound, triclosan, may be of limited use in household products it has been used in — soaps, deodorants, shaving creams, cosmetics and dishwashing liquids — and it is not removed during waste water treatment (Schwarcz, 2014.). Triclosan has been in wide use for forty years, and traces can be found in the urine of a majority of the US population (Calafat, Ye, Wong, Reidy, Needham, 2007.). Ironically, this product, intended to protect our health, may be jeopardizing it by disrupting the human microbiome, to say nothing of the effects of triclosan on fish and other aquatic life (Schwarcz, 2014.). Many governments are contemplating banning the substance (CBC News, 2014.), but this is but one example of our fear and ignorance of microbial life gone awry.
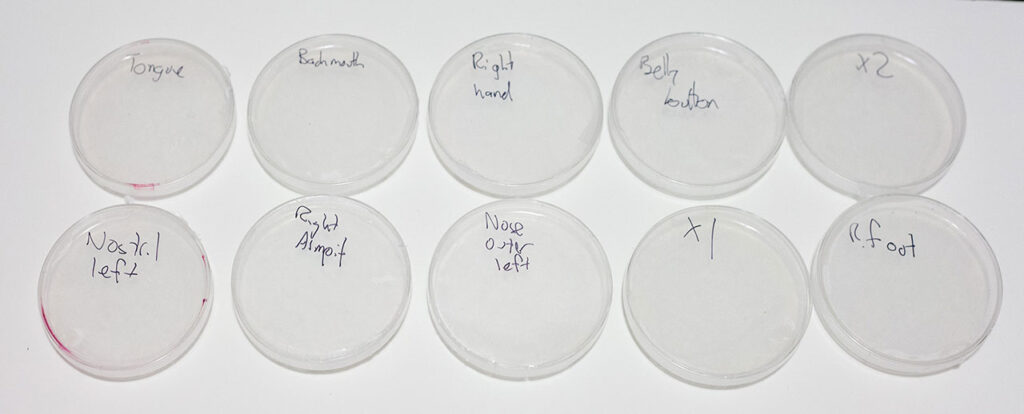
Once I finally got the petri dishes back to my house, I realized I did not have an ideal place to house them while countless thousands lived, underwent repeated mitosis, and died. For want of a proper incubator, I wrapped my ten petri dishes up in a towel and put them on a shelf above a baseboard heater. The thermometer I put in there regularly read between 25 and 30ºc, which is not ideal, but should have been sufficient to allow for slow growth. After a week, over the course of which I alternated between obsessively checking the dishes and forced indifference, I decided to call it off. Only two dishes — from the back of my mouth and my tongue (right) — had registered any change at all. I had hoped for horrendous, vivid, spreading growth, but had to settle for a few shiny bubbles. Even actively trying to get to know these microscale creatures, using expert, if borrowed, equipment yielded next to no results. My friend had counseled me that the most likely outcome was excessive growth, but with no sure way to tell what was ‘mine’ and what was environmental.

Ten sample dishes after seven days at ~25ºc.
I decided to attempt to create colonies of my own microbiota again, but this time using somewhat more rudimentary equipment. (see right) I carefully sterilized the previous colonies with a 10% bleach solution, thoroughly rinsed the dishes with previously boiled water, then heated up an agar ‘preparation’ that I’d bought in an Asian grocery store. Streaking the dishes with Q-tips from a freshly opened travel-size box, I doubted whether I’d actually culture anything that lived on me, or just whatever happened to live in my kitchen, or in the Q-tip wrapping machine. Nonetheless, I was hoping for something colourful and interesting. I’d even found a more reliably warm spot to incubate the dishes, as my landlord had finally fixed the heat in my apartment. 35ºc around the clock should have these dishes thriving in no time.

Definitely not a laboratory.
Or so I thought. After a week nothing had happened. Not even the standard-issue festering that I’d expect from something that I’d left near a radiator for seven days. It seemed that my own microbiome would remain entirely out of my reach.
Ten sample dishes after eight days at ~35ºc.
It is difficult to get to know our microbiota. Ingrained cultural mores around hygiene and cleanliness, legitimate fears of disease and infection, and simple matters of scale have long stymied any attempts to come to grips with our status as entire ecosystems instead of mere individual animals. While science has only recently begun to investigate our corporeal biomes in earnest, it is not too soon to question some of the traditional modes of thought which have given rise to some of the problematic, simplistic, and downright wrong ways of thinking about microbial life. I remain hopeful that the combination of a more measured, informed microbial epistemology and a thorough understanding of the form and function of our microbiomes through research endeavours like the Human Microbiome Project can provide us all with a future in which we can exist peacefully with our microbes, and interact with them in ways that preserve our health and theirs alike.
From the series ‘Micropolis’, Max Meyer, 2014.
References
- Bentley, R. & Meganathan, R. (1982). Biosynthesis of vitamin k (menaquinone) in bacteria. Microbiological Reviews, 46(3), 241-280.
- Bloomfield, S. F., Stanwell-Smith, R., Crevelz, R. W. R., & Pickup, J. (2006) Too clean, or not too clean: the hygiene hypothesis and home hygiene. Clinical and Experimental Allergy, 36, 402-425.
- Bourzac, K. (2007, July 26). Viewing bacterial dark matter. MIT Technology Review. Retrieved from http://www.technologyreview.com/news/408309/viewing-bacterial-dark-matter
- Calafat, A. M., Ye, X., Wong, L.-Y., Reidy, J. A., & Needham, L. L. (2007). Urinary concentrations of triclosan in the U.S. Population: 2003-2004. Environmental heath perspectives. Retrieved from http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2265044
- Canadian Press. (2014, July 10). Ban antibacterials triclosan and triclocarban, report says. CBC news. Retrieved from http://www.cbc.ca/news/technology/ban-antibacterials-triclosan-and-triclocarban-report-says-1.2703095
- Center for Disease Control and Prevention. (2014). Show me the science: When to use hand sanitizer. Handwashing: Clean hands save lives. Retrieved from http://www.cdc.gov/handwashing/show-me-the-science-hand-sanitizer.html
- DeLong, E. (2007, July 7). Metagemonics defined. MIT Technology Review, 26-27.
- Grice, E. A. & Segre, J. A. (2011). The skin microbiome. Nature Reviews Microbiology, 9, 244-253.
- Gut Flora. (n.d.). Retrieved November 28, 2014 from Wikipedia: http://en.wikipedia.org/wiki/Gut_flora
- National Institutes of Health. (n.d.). Human microbiome project: overview. Office of strategic coordination: The common fund. Retrieved from http://commonfund.nih.gov/hmp/overview
- Okada, H., Kuhn, C., Feillet, H., & Bach, J.-F. (2010). The ‘hygiene hypothesis’ for autoimmune and allergic diseases: an update. Clinical and Experimental Immunology, 160, 1-9.
- O’Keefe, S. J. D. (2008). Nutrition and colonic health: The critical role of the microbiota. Current Opinion in Gastroenterology, 24(1). Retrieved from http://www.medscape.com/viewarticle/568373_2
- Rosebury, T. (1969). Life on man. New York, NY: The Viking Press.
- Schloss, P. D. (2014). An integrated view of the skin microbiome. Nature, 514, 44-45.
- Schwarcz, J. (2014). Marketing appears to trump science on antibacterials. McGill office of science & society. Retrieved from http://blogs.mcgill.ca/oss/2014/07/24/marketing-appears-to-trump-science-on-antibacterials
- Skin Flora. (n.d.). Retrieved November 28, 2014 from Wikipedia: http://en.wikipedia.org/wiki/Skin_flora
- Smith, M. (2013). Ecological community, the sense of the world, and senseless extinction. Environmental Humanities, 2, 21-41.
- Strachan, D. P. (1989). Hay fever, hygiene, and household size. British Medical Journal, 299, 1259-1260.
- World Health Organization. (2009). Religious and cultural aspects of hand hygiene. WHO Guidelines on Hand Hygiene in Health Care. Retrieved from http://www.ncbi.nlm.nih.gov/books/NBK143998
Images
- Distribution of Microbe Species. Retrieved from http://www.genome.gov/dmd/img.cfm?node=Photos/Graphics&id=85320
- Images used in ‘Micropolis’ were originally taken by NASA’s Marshall Space Flight Centre and were released under the Creative Commons Attribution-NonCommercial 2.0 Generic License.